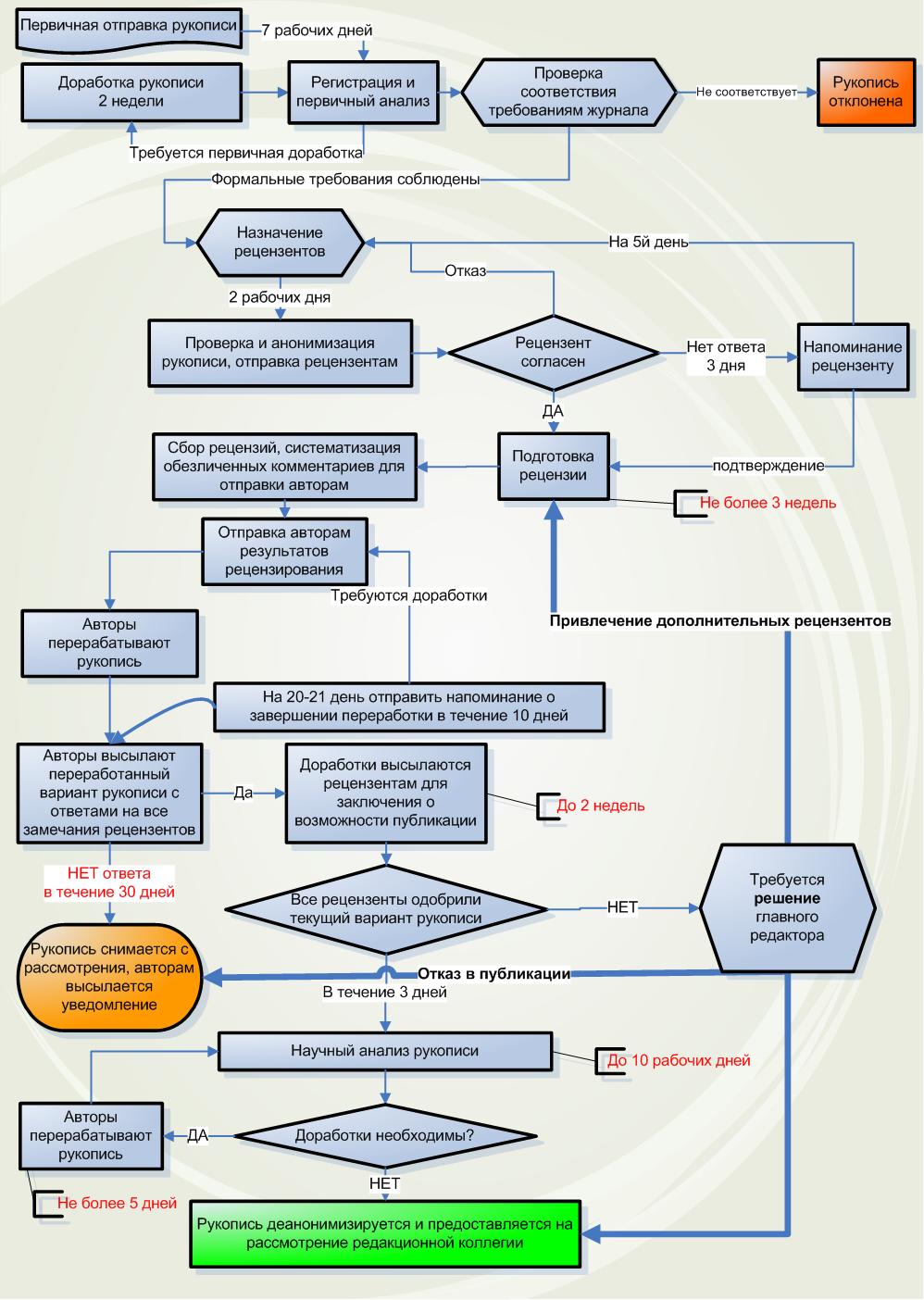
Основные принципы рецензирования
- Политика рецензирования призвана соответствовать лучшим практиками и этическим нормам, изложенным в рекомендациях COPE и ICMJE.
- Используется ДВОЙНОЕ СЛЕПОЕ рецензирование (рецензенты не знают идентификации авторов, включая их аффилиации, а авторы – рецензентов). Ослепление производиться ответственным секретарем.
- В отношении рукописей членов редколлегии также используется Двойное Слепое рецензирование.
- Авторы могут сами предложить рецензентов. Однако, окончательное решение о выборе того или иного рецензента принимает главный редактор.
- Рецензирование осуществляется членами редакционной коллегии и редакционного совета, а также приглашенными рецензентами – ведущими специалистами в соответствующей научной области России и других стран.
- Каждый рецензент имеет право отказаться от рецензии в случае наличия явного конфликта интересов, отражающегося на восприятии и интерпретации материалов рукописи.
- Каждую рукопись рассматривают минимум 3 рецензента.
- Рецензирование выполняется на безвозмездной основе.
- Рецензирование призвано установить соответствие рукописей, заявленным целям и задачам, а также предметно-тематической области журнала. Рукописи проверяются на предмет научной новизны и клинической значимости, отсутствия плагиата, корректности статистического анализа, четкости и понятности изложения, а также соответствию всех этических норм в сфере биомедицинских исследований. Среди других целей – поддержка прозрачности, воспроизводимости и обмена данными (включая должную регистрацию клинических исследований).
- Рецензирование проводится согласно внутренней форме и чек-листа рецензента и членов редколлегии, требующих развернутого аргументированного изложения, необходимой информации о сроках и условиях научного рецензирования, режиме конфиденциальности и защите персональных данных (включая GDPR) и пр.
- В рамках процедуры несогласий автора с решением редколлегии автор вправе направить письменную аргументированно-изложенную претензию (один раз в отношении одной рукописи). Редколлегия обязана ее рассмотреть не позднее 3-х недель и вынести окончательное решение, которое не подлежит пересмотру.
- Издательство и учредители делают все возможное для постоянной переподготовки редколлегии, а также проведения открытых семинаров для потенциальных авторов по вопросам биомедицинской этики, лучших мировых практик и рекомендаций. Журнал – член АНРИ (Ассоциации научных редакторов и издателей, аффилированной с European Association of Science Editors), которая по запросу Журнала проводит его аудит на соответствие международным и национальным стандартам публикационный этики и лучшим мировым практикам.
См. дополнительную информацию об этической политике и практике в отношении нарушений в области рецензирования, а также взаимоотношений с редколлегией и рецензентами в соответствующем разделе.
Ключевые сроки и иные показатели
- Процент Принятия/Отказа рукописей: 58/42% (включая 24% отказа на первоначальном этапе скрининга до назначения рецензирования)
- Срок первоначального рассмотрение/ скрининга рукописи: не более 10-ти дней
- Срок от получения рукописи до принятия в печать: в среднем не менее 62-х дней
- Срок хранения рецензий: 5 лет
Схема маршрутизации (рецензирования) рукописей